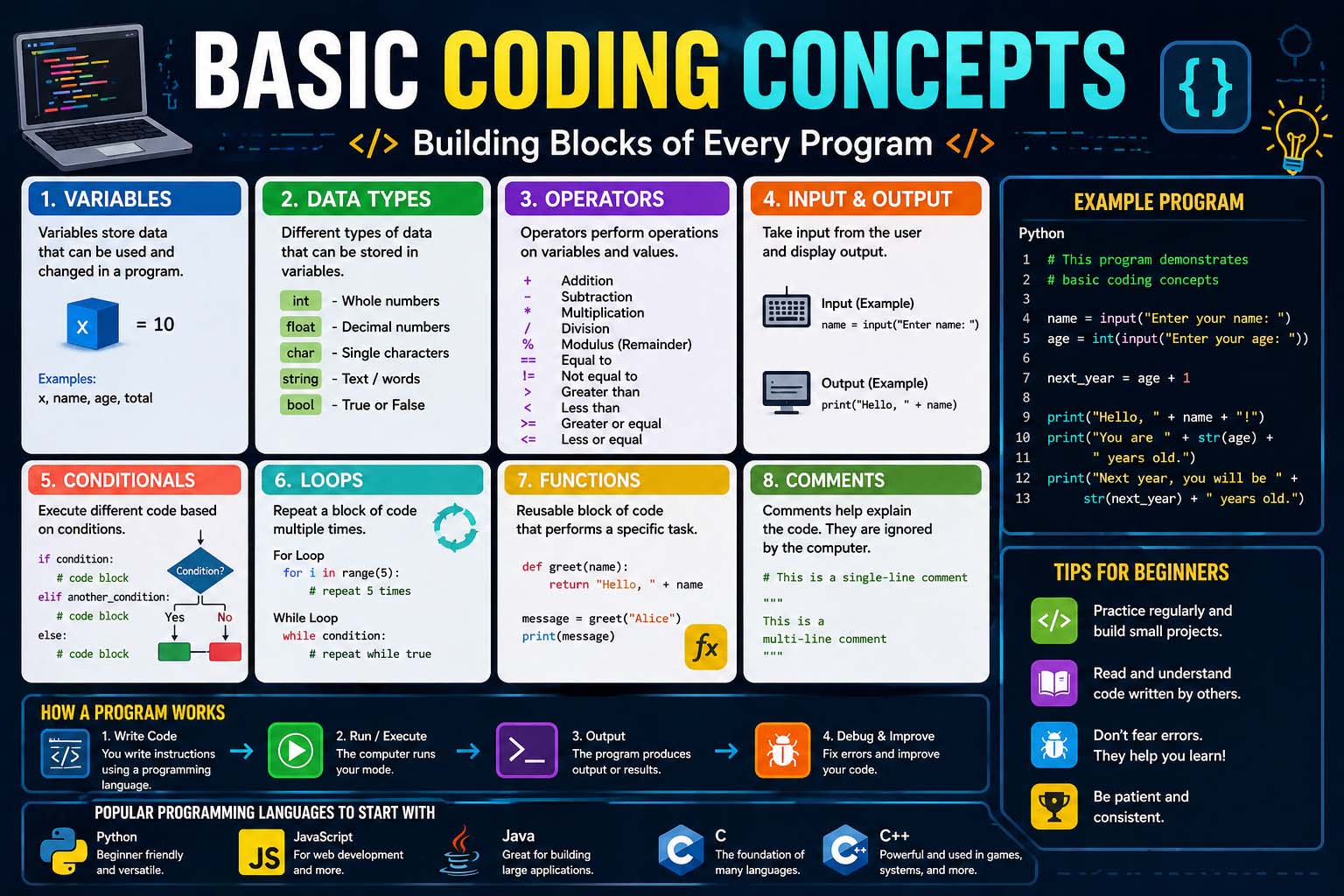
Understanding the physical change definition is an important part of learning basic science and chemistry. A physical change happens when the form, size, shape, or state of matter changes without creating a new substance. In simple words, the material may look different, but its chemical identity remains the same.
Physical changes are common in everyday life. Ice melting into water, paper being cut into smaller pieces, and sugar dissolving in tea are all examples. These changes are easy to observe and help students understand how matter behaves.
Learning about physical changes helps build a strong science foundation because it teaches the difference between changing appearance and changing composition.
What Is a Physical Change?
If you ask what is the definition of physical change, the answer is straightforward. A physical change is any change that affects the physical properties of a substance without changing what the substance is made of.
Properties that may change include:
- Size
- Shape
- Texture
- Color appearance
- State of matter
- Volume
Even after the change, the substance is still the same material. For example, water can freeze into ice or boil into steam, but it remains H2O.
Definition of Physical Change
The definition of physical change in science refers to a transformation where no new substance is formed. The molecules may move farther apart, come closer together, or rearrange physically, but they do not become different molecules.
This concept is useful because it separates physical changes from chemical changes. In a chemical change, atoms rearrange to create something new. In a physical change, the substance keeps the same identity.
For students, remembering “same substance, different form” is a helpful shortcut.
Physical Change Definition Chemistry
The physical change definition chemistry often focuses on how matter changes state or form while keeping the same chemical formula.
For example:
- Ice melting into liquid water
- Water evaporating into steam
- Aluminum foil being folded
- Salt dissolving in water
- Wood being cut into pieces
Chemistry studies these changes to understand energy, particle movement, and the properties of matter.
Even when a material changes shape or phase, its chemical makeup remains unchanged.
Physical Change Science Definition
The physical change science definition is commonly taught in classrooms to help explain observable changes in matter. Science uses physical changes to demonstrate how temperature, pressure, and force can affect materials.
Examples include:
- Crushing a can
- Stretching rubber
- Breaking glass
- Tearing paper
- Melting wax
These examples show that matter can change appearance without changing into a different substance.
Common Examples of Physical Changes
Physical changes happen constantly in homes, schools, and nature. Some of the easiest examples include melting ice cubes, freezing water, and boiling water for cooking.
Cutting vegetables changes their size and shape but not their identity. Sharpening a pencil changes its form. Inflating a balloon changes size because air is added.
Dissolving sugar in water is another common example. The sugar seems to disappear, but it is still present and can often be recovered by evaporation.
Signs of a Physical Change
Recognizing a physical change becomes easier when you know the common signs. These often include:
- Change in shape
- Change in size
- Change in state
- Dissolving
- Breaking apart
- Mixing without reaction
Unlike chemical changes, physical changes usually do not produce gas, permanent color change, or a completely new substance.
However, some observations may overlap, so careful testing is sometimes needed.
Physical Change vs Chemical Change
Many students confuse physical and chemical changes. The main difference is whether a new substance forms.
Physical change:
- No new substance created
- Often reversible
- Changes appearance or state
Chemical change:
- New substance formed
- Often difficult to reverse
- May involve burning, rusting, bubbling, or odor change
For example, melting butter is a physical change, while burning butter is a chemical change.
Understanding this difference is essential in science education.
Is a Physical Change Reversible?
Many physical changes can be reversed. Ice melts into water, and water can freeze again. Steam can cool back into liquid water.
A paper sheet torn into pieces is harder to restore, but the material is still paper. So while not all physical changes are easily reversible, many can be undone through physical means.
Reversibility is a clue, but it is not the only factor used to identify physical change.
Why Physical Changes Matter
Physical changes are important in science, manufacturing, cooking, and engineering. Many industries rely on shaping, cutting, freezing, heating, or compressing materials without altering chemical composition.
Examples include:
- Recycling metals by melting and reshaping
- Freezing foods for storage
- Cutting fabric in clothing production
- Molding plastic products
- Crushing minerals into powder
These processes help create useful products efficiently.
How to Teach Physical Change to Students
Teachers often use hands-on experiments to explain physical changes. Melting ice, tearing paper, or dissolving salt in water are simple classroom demonstrations.
Students learn better when they can observe matter directly. Comparing physical and chemical changes side by side also improves understanding.
Using everyday examples makes the concept easier to remember.
Conclusion
The physical change definition describes a change in matter where the substance keeps the same identity while its form, shape, or state changes. From melting ice to cutting paper, physical changes are part of everyday life.
Whether studying the definition of physical change, learning the physical change definition chemistry, or reviewing the physical change science definition, the key idea remains simple: the material changes appearance, not composition.
Understanding physical changes helps students build a strong foundation in science and better understand the world around them.