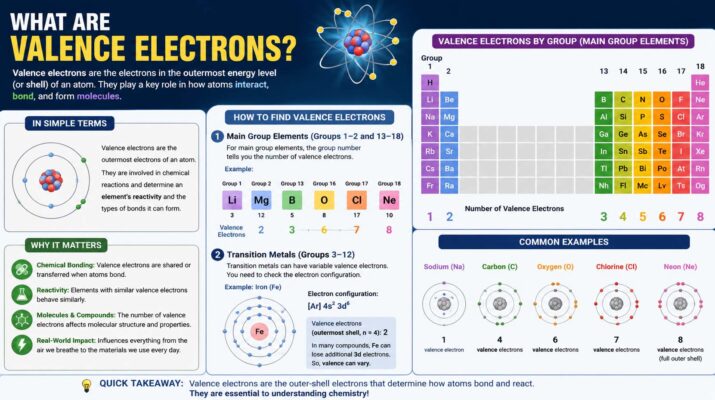
Valence electrons are one of the most important concepts in chemistry because they explain how atoms interact with each other. Every atom contains electrons that move around the nucleus in different energy levels or shells. The located in the outermost shell are called valence electrons. These electrons play a major role in chemical bonding, reactions, and the overall behavior of elements.
Understanding what are valence electrons helps students and science enthusiasts learn why some elements react strongly while others remain stable. Elements with full outer shells are usually less reactive, while those with incomplete shells tend to form chemical bonds more easily. From simple table salt to complex biological molecules, valence electrons influence nearly every chemical process in nature.
What Are Valence Electrons?
The question “what are ” is often one of the first topics introduced in chemistry classes. These electrons are involved in forming bonds with other atoms and determine how an element behaves chemically.
. The electrons orbit the nucleus in layers called shells. Inner-shell electrons remain close to the nucleus and usually do not participate in chemical reactions. However, the outermost electrons are more loosely held, which allows them to interact with electrons from other atoms.
For example, sodium has one valence electron in its outer shell. Because it has only one outer electron, sodium easily loses it during chemical reactions. Chlorine, on the other hand, has seven and needs one more to complete its shell. When sodium and chlorine combine, sodium gives its electron to chlorine, forming table salt.
The number of valence electrons also helps scientists predict how elements will react. Elements in the same group of the periodic table usually have the same number of valence electrons, which explains why they often share similar chemical properties.
How Valence Electrons Affect Chemical Bonding
Chemical bonding occurs because atoms seek stability. Most atoms become stable when their outer shell contains a certain number of electrons, often eight. Valence electrons are directly responsible for this process because they are exchanged or shared between atoms.
There are two major types of chemical bonds influenced by . Covalent bonds form when atoms share electrons. Both processes depend entirely on the arrangement of valence electrons.
For instance, oxygen has six and needs two more for stability. Hydrogen has one valence electron and needs one more. When oxygen bonds with two hydrogen atoms, they share electrons and create a water molecule. Without , chemical compounds like water, carbon dioxide, and proteins could not exist.
What Are Valence Electrons and Why Are They Important
Many students ask, “what are and why are they important?” The answer lies in their role in chemistry, biology, and even modern technology. control how atoms bond, how compounds form, and how substances react with one another.
In biology, are essential because they help create the molecules necessary for life. Carbon, hydrogen, oxygen, and nitrogen all depend on electron sharing to form proteins, DNA, carbohydrates, and other biological structures. Every living organism relies on chemical bonds created by valence electrons.
In industrial chemistry, valence help scientists design medicines, fuels, plastics, and fertilizers. Understanding electron behavior allows chemists to predict reactions and create safer, more effective products. Electronics and batteries also depend on the movement of to generate and store energy.
Valence electrons are important in environmental science as well. Chemical reactions in the atmosphere, soil, and oceans occur because of electron interactions. Scientists studying pollution or climate processes often examine how atoms exchange or share during reactions.
What Are the Valence Electrons
When asking “what are the ,” it is helpful to look at examples from the periodic table. Each element has a specific number of that determines its chemical behavior.
Hydrogen has one valence electron. Carbon has four. Oxygen has six, while neon has eight. Elements with nearly full or nearly empty outer shells are usually the most reactive because they seek stability through bonding.
The periodic table makes it easier to identify . For many main-group elements, the group number corresponds to the number of valence electrons. Group 17 elements like chlorine and fluorine have seven
Noble gases in Group 18 already have full outer shells, making them highly stable and less likely to react. This stability explains why gases like helium and neon are chemically inactive compared to more reactive elements.
What Are Valence Shell Electrons
The phrase “what are valence shell ” refers to the same outermost involved in chemical bonding. The valence shell is simply the atom’s outermost electron shell, and the electrons inside it are known as shell electrons.
These electrons are farther from the nucleus than inner-shell electrons, which makes them easier to gain, lose, or share during reactions. Because of this position, valence shell electrons have the greatest influence on an atom’s chemical properties.
For example, magnesium has two valence shell . It tends to lose both electrons during reactions to achieve a stable electron arrangement. Sulfur has six valence shell and often gains two to complete its outer shell. These predictable behaviors are based entirely on valence shell electron arrangements.
Conclusion
Learning what are electrons is essential for understanding chemistry and the behavior of matter. These outermost electrons determine how atoms bond, react, and form compounds. From water molecules to advanced electronic devices, influence countless processes in science and daily life.